Web developers, researchers and the public will now have easier access to FDA public health datasets thanks to the US regulator’s new open data initiative.
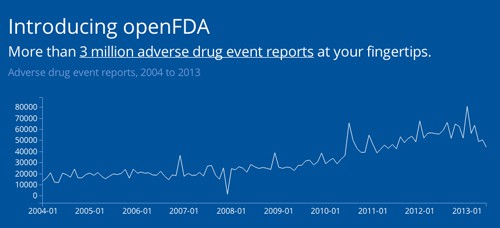
The openFDA project began this week with the release of millions of reports of drug adverse events and medication errors that have been submitted to the FDA between 2004 and 2013.
The data had previously been available, but only through a difficult-to-use reporting system or via Freedom of Information Act requests.
Taha Kass-Hout, the FDA’s chief health informatics officer, said: “Through this new and novel approach to data organisation, these reports will be available in their entirety so that software developers can build tools to help signal potential safety information, derive meaningful insights, and get information to consumers and healthcare professionals in a timely manner.
“OpenFDA offers a scalable platform that can be easily searched and queried across many distinct datasets, and can be easily redeployed or altered to fit a variety of purposes, and provides an innovative public data search and analytics solution.”
The first release of data will followed by the FDA’s databases on product recalls and product labelling, with the whole project coming in the way of the Presidential Executive Order on Open Data and the Department of Health and Human Services’ Health Data Initiative.
The openFDA website makes the FDA’s publicly available data accessible in a structured, computer readable format through a search-based application program interface (API).
The regulator says this will make it possible for the likes of mobile application creators, web developers, data visualisation artists and researchers to quickly search and make use of public information contained in the FDA’s datasets.




