PharmaQuest returns: EPA perspectives, through a rare disease lens
April 7, 2026 | EPA, First‑in‑class innovation, PharmaQuest, Quality of life (QoL), ispor, rare diseases
By Lottie Sequerra and Emily Morton-West

At ISPOR 2025, we launched our interactive survey, PharmaQuest, challenging stakeholders to make real-world trade-offs in designing the ‘ideal’ therapy (see our ISPOR results blog here). When PharmaQuest returned at the World Evidence, Pricing and Access (EPA) Congress 2026, we examined whether the priorities seen at ISPOR also held true for an EPA audience. The results reveal a striking consistency across conferences, with a particular emphasis on attributes relevant to rare diseases, such as unmet need, first-in-class innovation, and improvements in quality of life (QoL).
What priorities aligned across EPA and ISPOR stakeholders?
- First-in-class therapies
- Treatments targeting low-prevalence populations with high unmet need
- Selected profiles offering moderate efficacy with significant improvements in QoL
Together, these shared preferences point to an aligned set of decision-making principles that extend across stakeholder groups and conference settings.
Rare diseases take centre stage at EPA
The most popular therapy areas varied between congresses. ISPOR respondents marginally prioritised oncology over rare diseases, while EPA attendees showed a clearer preference for rare disease therapies. This may in part reflect the timing of EPA, which occurred shortly after a wave of policy and guidance activity across key markets. This included new and updated frameworks such as the European Added Value Assessment examining the case for a more coherent EU‑level framework for rare diseases, as well as NICE’s new quality standard for rare diseases with statements on access to treatment. This emphasis was reinforced at EPA, which featured a dedicated rare diseases track highlighting the importance of involving patients early to ensure evidence generation focuses on outcomes that matter to patients, reflects lived experience, and supports timely access to treatment. More broadly, regulatory and market access discussions around mechanisms such as FDA priority review vouchers and the EU pharmaceutical package, including extended market exclusivity for orphan and high‑unmet‑need breakthrough therapies, have kept rare diseases firmly at the forefront of market access stakeholder priorities.
Summary of responses to the question, “Select the therapy area you would prioritize for designing your ideal drug”
Therapy areas prioritized for ideal drug design at EPA

Therapy areas prioritized for ideal drug design at ISPOR

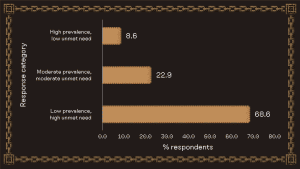
Do EPA stakeholders prioritize unmet need over prevalence?
The focus on rare diseases at EPA was mirrored by respondents showing a strong preference for therapies targeting low-prevalence populations, with high unmet need over the alternative options. With only a minority of rare diseases currently having an approved treatment, these results reflect a clear willingness among stakeholders to prioritize areas where therapeutic options remain limited, and the potential for meaningful impact is greatest.
Summary of responses to the question, “Would you prioritize an indication with high prevalence or high unmet need?”
First‑in‑class innovation a clear priority at EPA
Around three-quarters of respondents prioritized a first‑in‑class therapy at EPA when designing their ‘ideal’ drug. Such therapies are extremely common in rare disease innovation, where novel mechanisms of action are often required to address previously untreated or poorly understood conditions. Notably, this preference was unanimous among healthcare professionals and payers, while a minority of pharmaceutical respondents selected therapies with an established mechanism of action. This divergence may reflect varying perspectives on development risk, with established mechanisms often offering greater predictability, clearer unmet need narratives, and more mature epidemiological or registry data. Considerations around development feasibility and infrastructure, including experience from complex areas such as cell and gene therapies, may also shape these preferences.
Summary of responses to the question, “Would you prefer a first-in-class product offering higher risk or an established mechanism with lower risk potential?” stratified by respondent type

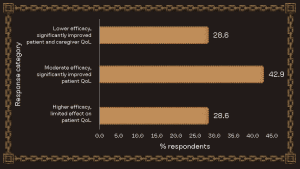
The preferred efficacy profile at EPA
The most selected efficacy profile at EPA favoured moderate clinical efficacy combined with a significant improvement in QoL (quality of life). This aligns closely with the realities of rare disease development, where many existing treatments may not address the root cause of the disease; however, substantial value can still be delivered through improvements in daily functioning, symptom burden, and long-term disease management.
Summary of responses to the question, “Would you prioritize higher efficacy over better QoL?”
Are stakeholders willing to trade cost-effectiveness for curative potential?
At EPA, preferences were more evenly split compared to other PharmaQuest questions when respondents were asked: “Would you prefer a product that is curative but exceeds cost-effectiveness thresholds or one that treats symptoms and is significantly more affordable?” 48.6% of respondents chose a curative treatment that was less cost-effective (incremental cost-effectiveness ratio [ICER] above accepted thresholds), and 51.4% of respondents selected a treatment that relieves symptoms but is more cost-effective (ICER below accepted thresholds). However, clear differences emerged when responses were analysed by role. 75% of healthcare professionals showed a stronger preference for symptom-relieving therapies that were more affordable, while 75% of pharmaceutical respondents prioritized curative approaches that were less cost-effective. For healthcare professionals, this may reflect the lived clinical experience of the impact that symptom management can deliver for patients, particularly where improvements in daily functioning can be highly meaningful even in the absence of a cure, and where budget constraints remain a practical consideration. In contrast, pharmaceutical preferences may reflect pipeline and portfolio decision‑making considerations, with a focus on advancing the most efficacious candidates and a greater willingness to tolerate uncertainty in cost-effectiveness metrics. This approach is often seen in rare disease development, where high unmet need and limited alternatives can justify greater ICER uncertainty.
What does this mean for market access strategy?
Taken together, findings from PharmaQuest at EPA and ISPOR highlight a consistent set of priorities shaping market access decision-making, with attributes relevant to rare disease repeatedly emerging across stakeholder groups and settings. Preferences for first‑in‑class innovation, therapies addressing low‑prevalence conditions with high unmet need, and value delivered through meaningful improvements in QoL underscore a shared recognition that impact extends beyond traditional measures of efficacy alone. While contextual factors and audience composition can influence how trade-offs are weighed, the alignment observed suggests a stable and enduring focus on addressing unmet need when defining value in today’s evolving market access landscape. In this environment, having a compelling unmet need narrative and clearly defined positioning becomes especially important amid ongoing shifts in pharmaceutical policy and broader uncertainty. Together, these factors reinforce how value communication must stay aligned with stakeholder priorities and continue evolving alongside the rapidly changing access landscape.
For more insights, visit our website
This content was provided by Amiculum





