From text to context: Helping patients see side effects clearly
March 30, 2026 | health information, health literacy, healthcare communications
Discover how thoughtful design, clear language, and structured visuals can transform overwhelming side effect information into accessible, actionable insights for patients in our latest Health Literacy series.

Our Health Literacy series continues, and this time we’re sharing another example of how thoughtful design, structure and language are essential to inform, educate and empower patients — this time in identifying side effects.
If you have read a patient insert for a medicine, or an informed consent form, you will know that side effects lists are often long, confusing, and intimidating. This can make keeping track of what to look out for, and what to do about it, completely overwhelming.
But it doesn’t have to be. For example, our ‘before’ and ‘after’ examples below include the same information, presented in very different ways. And it’s often this that makes the greatest difference to patients: how clearly and accessibly information is communicated.
Before:

Safety information for patients included a dense, unformatted list of all potential side effects. The descriptions relied heavily on technical terminology, with no context, categories, or cues, making it difficult for readers to take away anything actionable.
After:
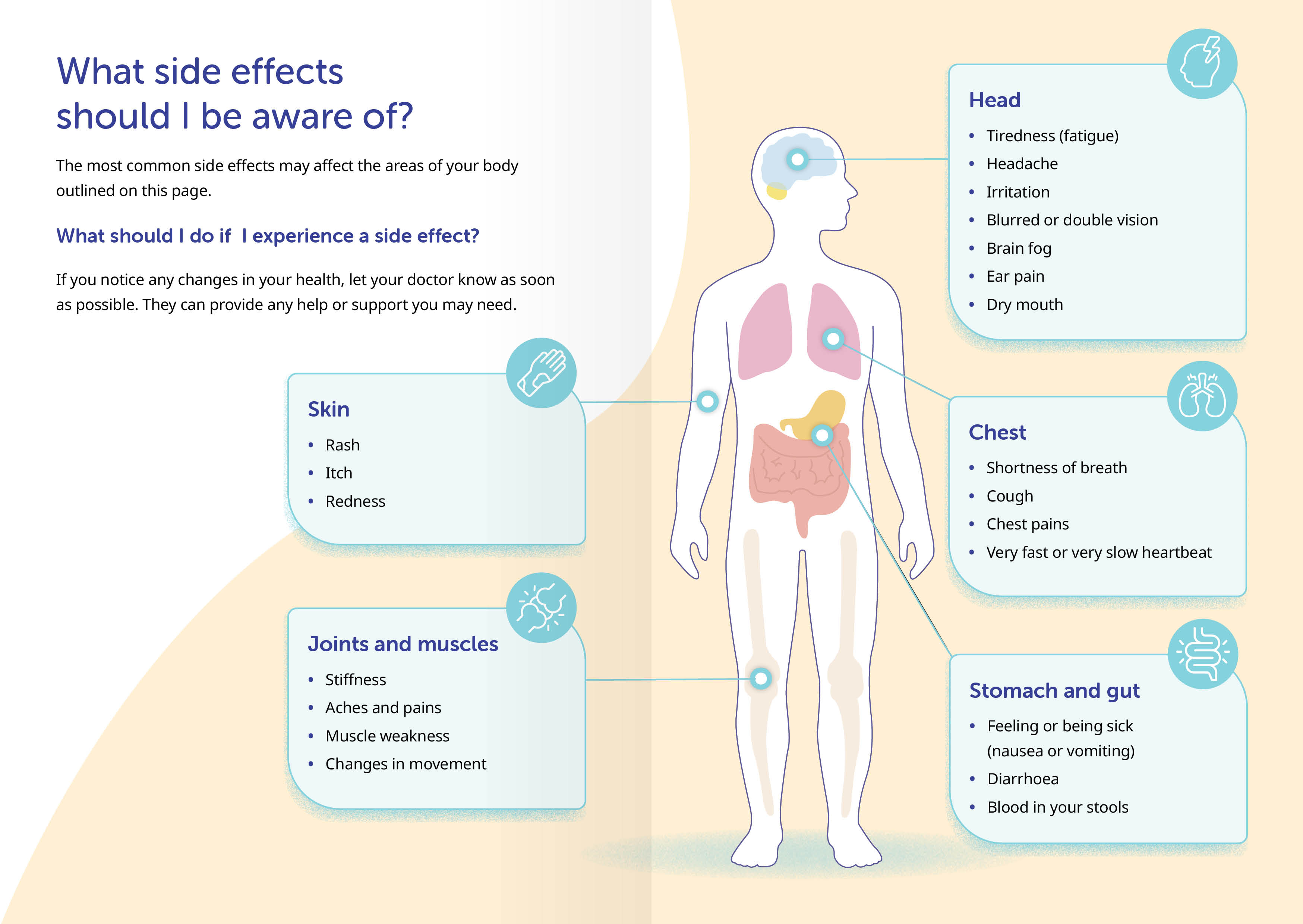
First, we translated all of the technical terms into lay language — “dry mouth” is just easier to process than “xerostomia”.
Next, we grouped everything by body system, so there’s no need to jump around the list for all related side effects.
Finally, we visualised each category on a silhouette, so readers know, at a glance, which body areas might be affected, and which specific changes to look out for.
By combining clear language and intuitive visuals, we’ve made potentially daunting side effect information easier to understand, recognise, and act on.
This content was provided by Cuttsy + Cuttsy
Company Details
Latest Content from Cuttsy + Cuttsy
Caroline Benson and Colin Williams to Chair the PM Society from February 2020
For the first time both industry and agencies will be equally represented as the PM Society appoints a joint chair.
Clinical trials, Actually.
This Christmas, we’re thinking of the thousands of clinical trial volunteers across the UK, both patients and healthy participants. We have a lot to thank them for...
A patient view on the ABPI’s patient collaboration sourcebook
Last month, the ABPI released a much-in-demand document: The Working with Patients and Patient Organisations sourcebook.
The Exhibition Hall: Past, Present and Future
As I sit here at another pharma conference, I start to reflect on how they have changed over the years.
Cuttsy+Cuttsy awarded CPD Platinum by the IPA
Four years after being awarded Gold for their continuous professional development (CPD) Cuttsy+Cuttsy (C+C) have reached another milestone and been awarded Platinum accreditation.
Clinical Trials Materials: Getting the Basics Right
For those unfamiliar with them, clinical trials can be rather bewildering. This presents problems when it comes to recruiting patients — how can we make sure they understand what they...
Cuttsy+Cuttsy named one of the Best Places to Work 2019 by Campaign
11 March 2019—Cuttsy+Cuttsy are honoured and extremely proud to be named one of the 50 Best Places to Work in 2019 by Campaign. The results were announced on 6th March...
Rare Disease Day 2019
Today is #RareDiseaseDay and is an opportunity to highlight some truly unbelievable/frightening statistics around rare diseases.
Health Literacy in Practice: Doctor’s Letter
If a patient was given their results directly, how could we make it into something they could understand and, most importantly, act on?
International Childhood Cancer Day 2019
The number of new cases per year is shocking but the journey and the outlooks have vastly improved. We've pulled together an infographic to show just what we mean with...





