Focus on Rare Diseases
May 10, 2017 | Medical Communications, medical education, rare diseases, recruitment
As pharma becomes more focused on rare disease therapies, Lucid Group recognises that medical education agencies need to respond.
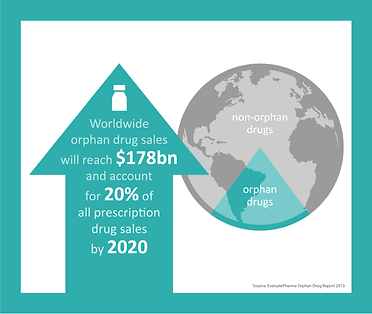
The US government passed the Orphan Drug Act in 1983, and it was subsequently adopted in other key markets, most notably in Japan in 1993 and the European Union in 2000. Growth in this sector has remained around 12% annually, which is more than double that experienced in the general drug market. According to the 2015 Orphan Drug Report, worldwide orphan drug sales will reach $178 billion by 2020 and will account for 20% of all prescription drug sales by 2020*.
The trend is clear: as science and technology advances, and the rise of the internet and social media enables patients and researchers to gather and share information more effectively, we are seeing treatments for rare diseases that only a few years ago would have led to an early death.
Alison Cantle, Rare Disease Business Unit Director at Lucid Group, explains what this means for medical education agencies: “As pharmaceutical organisations focus on personalised medicine, the need for rare disease expertise in their medical education agencies is paramount. Lucid recognised this and structured its business last year to create a unit specialising in rare diseases.”
Within this unit, Lucid Group had recruited a team of highly skilled medical writers who combine a deep and robust understanding of the science with experience of working with rare disease specialists across the globe.
“Our rare disease team understands the cornerstones of rare disease education and what makes it different from other therapy areas. It is essential to build networks and enable best practice sharing,” Alison explains. “And in rare disease, more so than in any other therapy area, the choice of agency is crucial, as often it is your agency who represents your organisation at the communication level – so its voice has to be your voice.”
Patients who have a rare disease face difficulty in every step of medical care, including diagnosis, treatment and preserving quality of life. Sometimes, patients go years without receiving the correct diagnosis for their condition. So raising awareness, improving time to diagnosis, getting patients to the right care, and improving family and emotional support and reimbursement are just some of the areas where medical education agencies can support pharma clients.
Lucid’s Rare Disease Business Unit currently works with clients in Fabry disease, Pompe disease, Gaucher disease, cystinosis and PKU. For further information about working with Lucid’s Rare Disease Business Unit, please call Alison on 01494 552078 or email alison@lucid-uk.com.
* EvaluatePharma Orphan Drug Report 2015
This content was provided by Lucid Group Communications Limited
Company Details
Latest Content from Lucid Group Communications Limited
Fiona’s #LucidLife
Meet Fiona, a medical writer.She told us what she wanted, and we did the rest.We believe that medical writers are like gold dust; so when we find talent, we do...
Lucid Group: What medical communications means to us
Katherine Duxbury, Scientific Director at Lucid, explains what medical communications means to Lucid: What we do, how we do it and what makes us different.
Lucid make the shortlist in all of the four categories they entered for the PMEA programme 2016
The PMEA programme rewards responsible customer-centric business practices that ensure treatments and advice provide patients with meaningful outcomes. The awards have been specifically designed to recognise and commend excellence, best...
Lucid bolsters its senior leadership team
Lucid Group has had a successful summer attracting top talent to its senior leadership team with the appointment of Shula Sarner (Executive Scientific Director) and Tanya Goodyear (Division Director, Immunology).
IBD Oxford MasterClass 2016 harnesses the chemistry between science and clinical practice
Leading Edge, part of the Lucid Group, delivered another successful Oxford MasterClass this year, which attracted over 260 delegates and 60 international inflammatory bowel disease (IBD) faculty, representing Europe, Asia, Australasia...
Lucid Group becomes a sponsor of MedComms Networking
To help Lucid access the valuable medical communications professional network they have become a sponsor of MedComms Networking, headed by Peter Llewellyn. Jan Steele (Co-founder and COO of Lucid Group)...
Tenth Lucid Academy module delivered – nurturing talent and driving excellence
Celebrating the success of the Lucid Academy
Lucid launch new recruitment campaign to attract top talent in medical writing
We will offer talented writers an exclusive, tailor-made package to suit their individual needs and desires. We want you to define the framework of your Lucid Life contract – from...
Knowing your audience is key to successful medical education
Lucid Group understands that the goals of medical education are to disseminate best practice and advance patient outcomes. Our approach to meeting these goals centres on our appreciation of both...
Making a difference together
It was highlighted at the 2016 World Economic Forum that ‘good business’ now means more than financial success, with multiple stakeholders asking how business will benefit society. At Lucid, our...