From text to context: Helping patients see side effects clearly
March 30, 2026 | health information, health literacy, healthcare communications
Discover how thoughtful design, clear language, and structured visuals can transform overwhelming side effect information into accessible, actionable insights for patients in our latest Health Literacy series.

Our Health Literacy series continues, and this time we’re sharing another example of how thoughtful design, structure and language are essential to inform, educate and empower patients — this time in identifying side effects.
If you have read a patient insert for a medicine, or an informed consent form, you will know that side effects lists are often long, confusing, and intimidating. This can make keeping track of what to look out for, and what to do about it, completely overwhelming.
But it doesn’t have to be. For example, our ‘before’ and ‘after’ examples below include the same information, presented in very different ways. And it’s often this that makes the greatest difference to patients: how clearly and accessibly information is communicated.
Before:

Safety information for patients included a dense, unformatted list of all potential side effects. The descriptions relied heavily on technical terminology, with no context, categories, or cues, making it difficult for readers to take away anything actionable.
After:
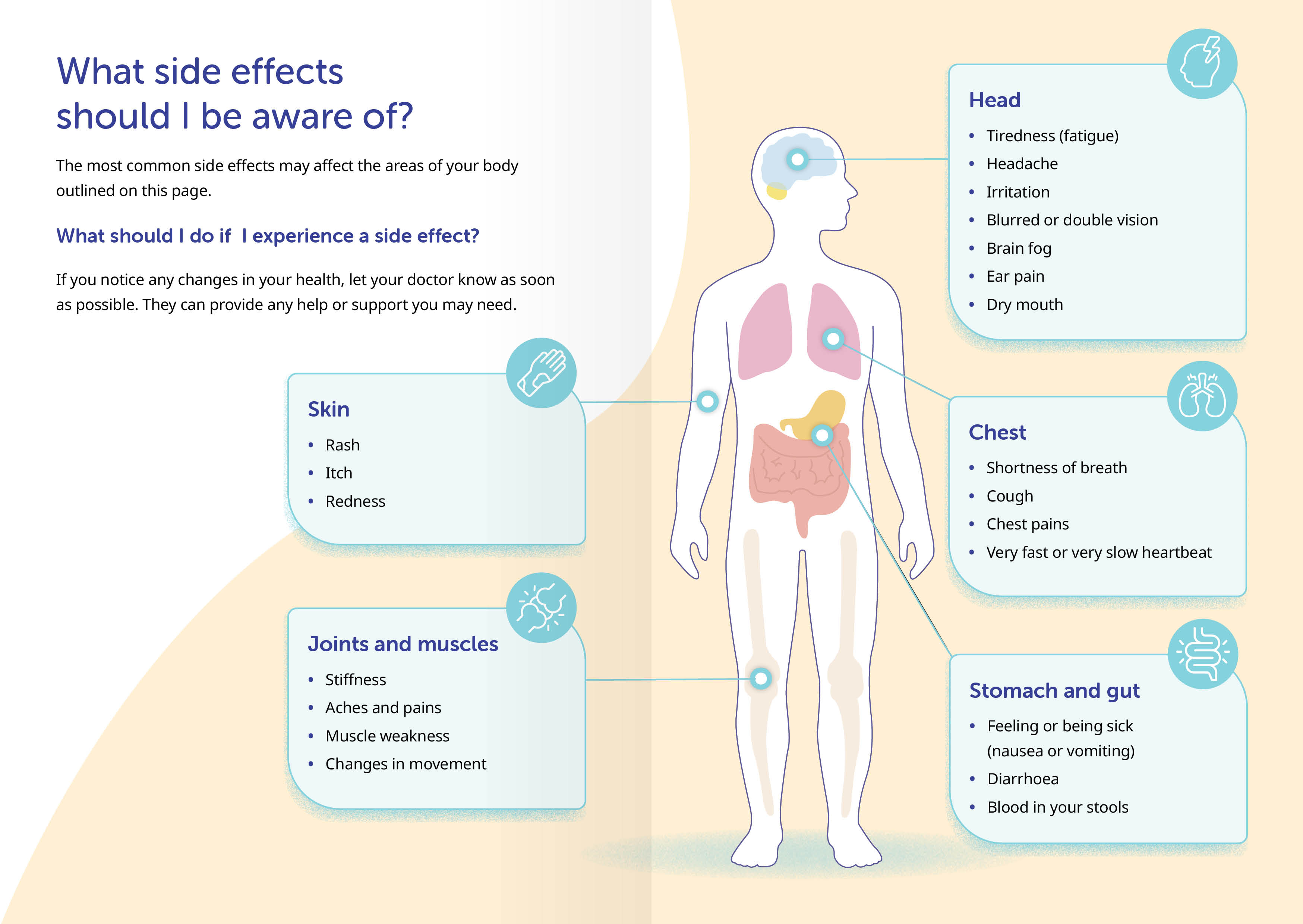
First, we translated all of the technical terms into lay language — “dry mouth” is just easier to process than “xerostomia”.
Next, we grouped everything by body system, so there’s no need to jump around the list for all related side effects.
Finally, we visualised each category on a silhouette, so readers know, at a glance, which body areas might be affected, and which specific changes to look out for.
By combining clear language and intuitive visuals, we’ve made potentially daunting side effect information easier to understand, recognise, and act on.
This content was provided by Cuttsy + Cuttsy
Company Details
Latest Content from Cuttsy + Cuttsy
How is generative AI transforming clinical trials?
Discover how generative AI is revolutionising clinical trials, from enhancing patient recruitment to improving data analysis.
Content for all – how to connect with neurodiverse audiences
Discover essential strategies for crafting inclusive and accessible written content tailored for neurodivergent readers.
Cuttsy+Cuttsy launches ccura: AI powered, always human
Cuttsy+Cuttsy introduces ccura, an AI-powered healthcare communications platform that blends technology with human expertise to enhance collaboration, streamline workflows, and deliver patient-focused, high-quality solutions with security and compliance at its...
Two Sides of the Same Coin: Patient Engagement vs Patient Involvement
Unpack the difference between patient engagement and patient involvement.
Cuttsy+Cuttsy achieves Platinum accreditation from Investors in People
Cuttsy+Cuttsy is proud to have achieved a Platinum accreditation from Investors in People, placing them among the top 2% of organisations assessed globally.
Cuttsy+Cuttsy awarded Gold EcoVadis sustainability rating
Cuttsy+Cuttsy proudly announces their Gold Medal achievement from EcoVadis for 2024, placing them among the top 5% of companies assessed and within the 97th percentile, a testament to their unwavering...
Age is not just a number when it comes to clinical trials
In this blog we look at the critical underrepresentation of older adults in clinical trials, emphasising the need for inclusive trial designs and targeted strategies to ensure safe and effective...
How to boost clinical trial access and make patients health-empowered
Clinical research is becoming more patient-centric, but there's still more that needs to be done to make clinical trials available to everyone. This could include wider participation.
Closing the loop: Why clinical trial results matter to participants
Discover why sharing clinical trial results with participants is crucial for respect, informed health decisions, and fostering strong researcher-participant partnerships.
Unveiling the truth: A journey into women’s representation in clinical trials
We explore the historical exclusion of women from clinical trials, its consequences on medical research and treatment efficacy and the ongoing efforts to ensure their representation.





