From text to context: Helping patients see side effects clearly
March 30, 2026 | health information, health literacy, healthcare communications
Discover how thoughtful design, clear language, and structured visuals can transform overwhelming side effect information into accessible, actionable insights for patients in our latest Health Literacy series.

Our Health Literacy series continues, and this time we’re sharing another example of how thoughtful design, structure and language are essential to inform, educate and empower patients — this time in identifying side effects.
If you have read a patient insert for a medicine, or an informed consent form, you will know that side effects lists are often long, confusing, and intimidating. This can make keeping track of what to look out for, and what to do about it, completely overwhelming.
But it doesn’t have to be. For example, our ‘before’ and ‘after’ examples below include the same information, presented in very different ways. And it’s often this that makes the greatest difference to patients: how clearly and accessibly information is communicated.
Before:

Safety information for patients included a dense, unformatted list of all potential side effects. The descriptions relied heavily on technical terminology, with no context, categories, or cues, making it difficult for readers to take away anything actionable.
After:
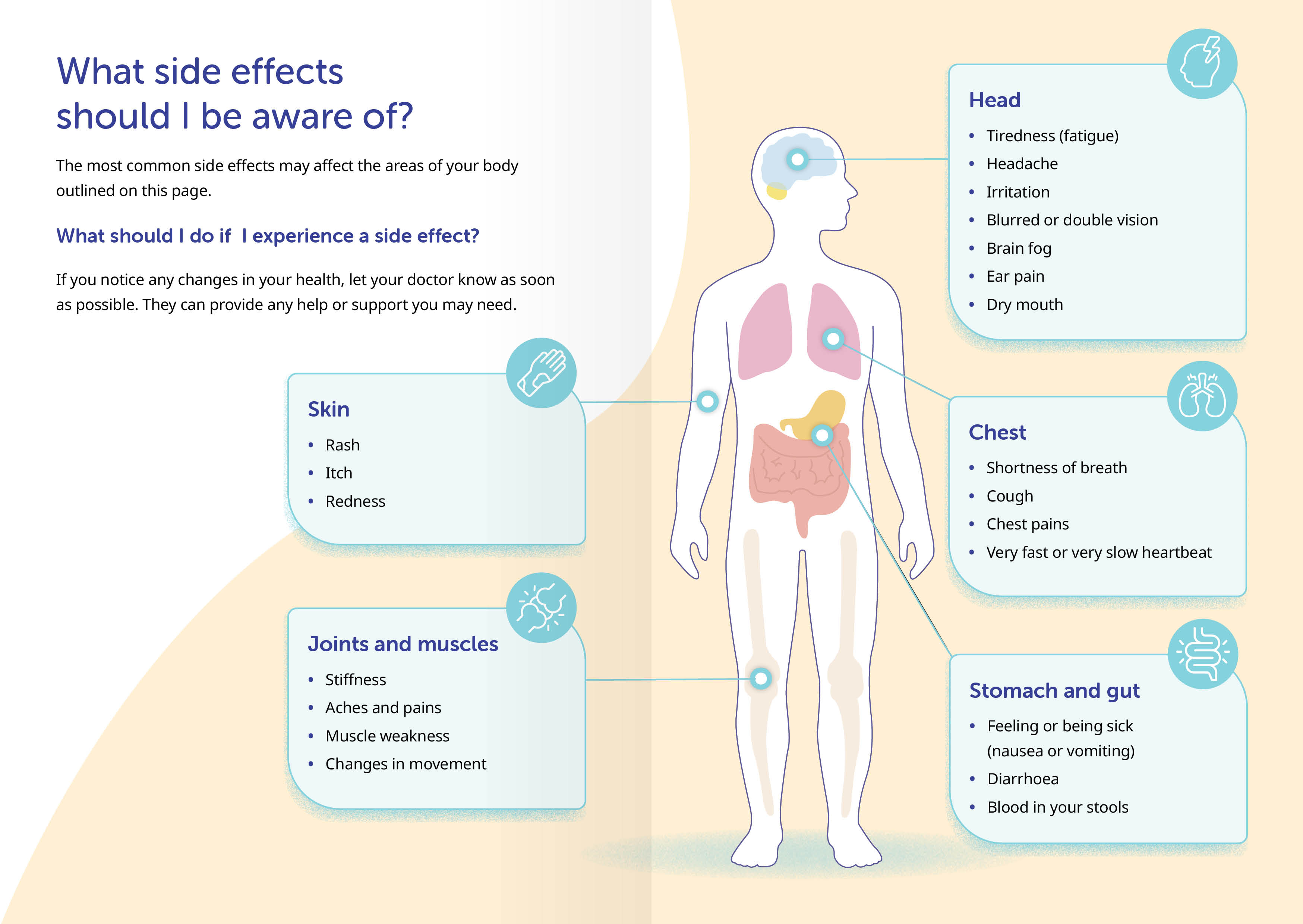
First, we translated all of the technical terms into lay language — “dry mouth” is just easier to process than “xerostomia”.
Next, we grouped everything by body system, so there’s no need to jump around the list for all related side effects.
Finally, we visualised each category on a silhouette, so readers know, at a glance, which body areas might be affected, and which specific changes to look out for.
By combining clear language and intuitive visuals, we’ve made potentially daunting side effect information easier to understand, recognise, and act on.
This content was provided by Cuttsy + Cuttsy
Company Details
Latest Content from Cuttsy + Cuttsy
Clinical trial considerations: Why diversity matters…
Diversity in clinical trials is crucial for understanding diseases and developing new treatments. Here's why diversity matters...
Clinical trial considerations: Why digital matters…
Digital technology in healthcare is rapidly evolving. In this blog we delve into how digital solutions can help improve patient experience within clinical trials.
Clinical trials explained: Why imagery matters…
Recruiting patients for clinical trials is one of pharma's greatest challenges. Imagery is a great way to connect with people on an emotional level and forms a big part the...
Clinical trials explained: Why health literacy matters…
Clinical trial recruitment can be a challenge. By applying the principles of health literacy to all materials, we can support people to make an informed choice about whether to take...
Clinical trials explained: Why iconography matters…
Icons serve as a useful form of alternative communication. We spend a lot of time developing icons for healthcare communications. Find out how we use iconography to elevate clinical trial...
Cuttsy+Cuttsy appoints Harriet Karia as Managing Director
Cuttsy+Cuttsy is pleased to announce the appointment of Harriet Karia as its first Managing Director.
Using generative AI in healthcare comms – our promise to our clients
It’s safe to say one of the hottest topics of 2023 has been ChatGPT and the launch of other Generative AI-driven applications. So, we’re sharing our promises, so that when...
Clinical trials explained: Why colour matters…
Colour can be used to emphasise mood, evoke emotions, and determine first impressions of a brand. Find how to use colour effectively in your next clinical trial campaign.
Clinical trials explained: Why branding matters…
With nearly a decade’s experience supporting clinical trials, find out why we think humanising trials through design is so important.
DPHARM2023 – Five key themes impacting clinical trials today
Throughout three fascinating days of conference, some key themes emerged that are impacting study teams and the effectiveness of their clinical trials on a daily basis.





