From cloudy to clear: visualising complex data privacy information
April 17, 2026 | clinical trials, communication, health literacy
Discover how clear communication principles can transform complex clinical research topics, as we conclude our Health Literacy series with a practical example on participant data transparency.

For the final instalment of our Health Literacy series, we’re sharing another example of how we apply clear communication principles in practice. Here, we explore how we approached a crucial topic in clinical research: participant data.
Medical data is private – it’s sensitive and not something you want to share with strangers. The lack of transparency around how clinical trial data is stored and used can be a major barrier to participation, particularly for underrepresented communities.
We understand the complexity that comes with this topic. There are so many regulations, nuances, and technicalities to consider, which often means the information reads like a Terms & Conditions page. Although the reader has been “informed”, have they really read it? And more importantly, do they really understand?
Our “before” and “after” example shows how a simple flow diagram can help to translate this critical information so anyone can understand how their personal data may be shared.
Before:

The language is contractual and uses technical terms withoutcontext. It’s difficult to know what data your doctor, a researcher, or thebroader scientific community might be able to see.
After:
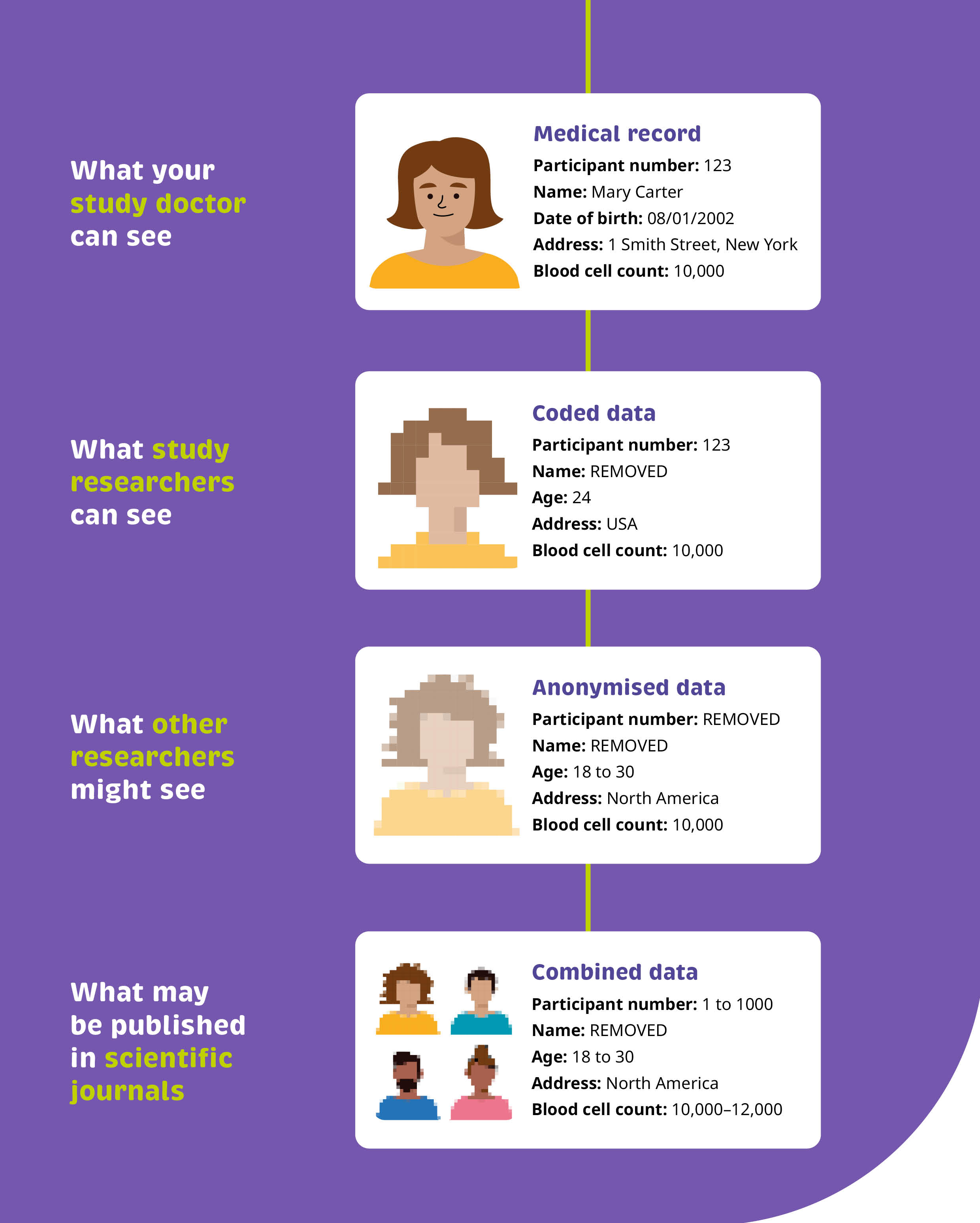
We’ve broken this information down into a flow diagram, clearly calling out the steps in place to ensure personal data are not identifiable. We made use of visual cues to emphasise that the data becomes more difficult to identify when it is processed and shared. Finally, using real examples of the types of data that may be collected helps provide the transparency readers are looking for.
Ultimately, we’ve provided a clear, visual commitment, supported with examples, to illustrate exactly how readers can expect their personal data to be used.
Clear communication about participant data isn’t just a regulatory requirement – it’s essential for building trust in clinical research. By presenting complex information in ways that people can easily understand, we can help more participants make informed decisions about taking part in clinical trials.
This content was provided by Cuttsy + Cuttsy
Company Details
Latest Content from Cuttsy + Cuttsy
Where it all began – why we developed The Experience Gap
Learn how The Experience Gap was developed to help sponsors, CROs and site teams improve clinical trial participation by identifying friction points, enhancing patient experience, and driving better recruitment and...
Cuttsy+Cuttsy launches a practical roadmap to enhance clinical trial participant experience
Cuttsy+Cuttsy has launched The Experience Gap, a practical roadmap designed to help clinical trial teams improve participant engagement, support and retention across the full study journey.
Cuttsy+Cuttsy welcomes three new experts to the team
Cuttsy+Cuttsy is happy to be welcoming three new people to the team this month.
The gap between hype and reality, what patients really say about weight loss drugs
Explore real patient conversations about weight loss drugs, uncovering challenges, support strategies, and the critical need for clear, reliable healthcare communication.
Why patient-centricity depends on site-centricity in clinical trials
Explore why patient-centricity in clinical trials relies on site-centricity, emphasising the need to design trials that empower site teams to better support patients.
From cloudy to clear: visualising complex data privacy information
Discover how clear communication principles can transform complex clinical research topics, as we conclude our Health Literacy series with a practical example on participant data transparency.
UK clinical trial regs are changing — here’s what’s happening
Discover the transformative updates to UK clinical trial regulations, effective from April 2026, and learn how they aim to enhance transparency, participant safety, and trial efficiency.
The invisible influence on trial retention: why caregivers must be part of the plan
Caregivers are vital to clinical trial retention, yet often overlooked—this article highlights their challenges, impact, and how to better support them.
Where it all began: why we wrote ‘The Site Factor’
Discover the story behind ‘The Site Factor’ and how understanding site teams’ experiences can transform clinical trials for participants and researchers alike.
From text to context: Helping patients see side effects clearly
Discover how thoughtful design, clear language, and structured visuals can transform overwhelming side effect information into accessible, actionable insights for patients in our latest Health Literacy series.





