From text to context: Helping patients see side effects clearly
March 30, 2026 | health information, health literacy, healthcare communications
Discover how thoughtful design, clear language, and structured visuals can transform overwhelming side effect information into accessible, actionable insights for patients in our latest Health Literacy series.

Our Health Literacy series continues, and this time we’re sharing another example of how thoughtful design, structure and language are essential to inform, educate and empower patients — this time in identifying side effects.
If you have read a patient insert for a medicine, or an informed consent form, you will know that side effects lists are often long, confusing, and intimidating. This can make keeping track of what to look out for, and what to do about it, completely overwhelming.
But it doesn’t have to be. For example, our ‘before’ and ‘after’ examples below include the same information, presented in very different ways. And it’s often this that makes the greatest difference to patients: how clearly and accessibly information is communicated.
Before:

Safety information for patients included a dense, unformatted list of all potential side effects. The descriptions relied heavily on technical terminology, with no context, categories, or cues, making it difficult for readers to take away anything actionable.
After:
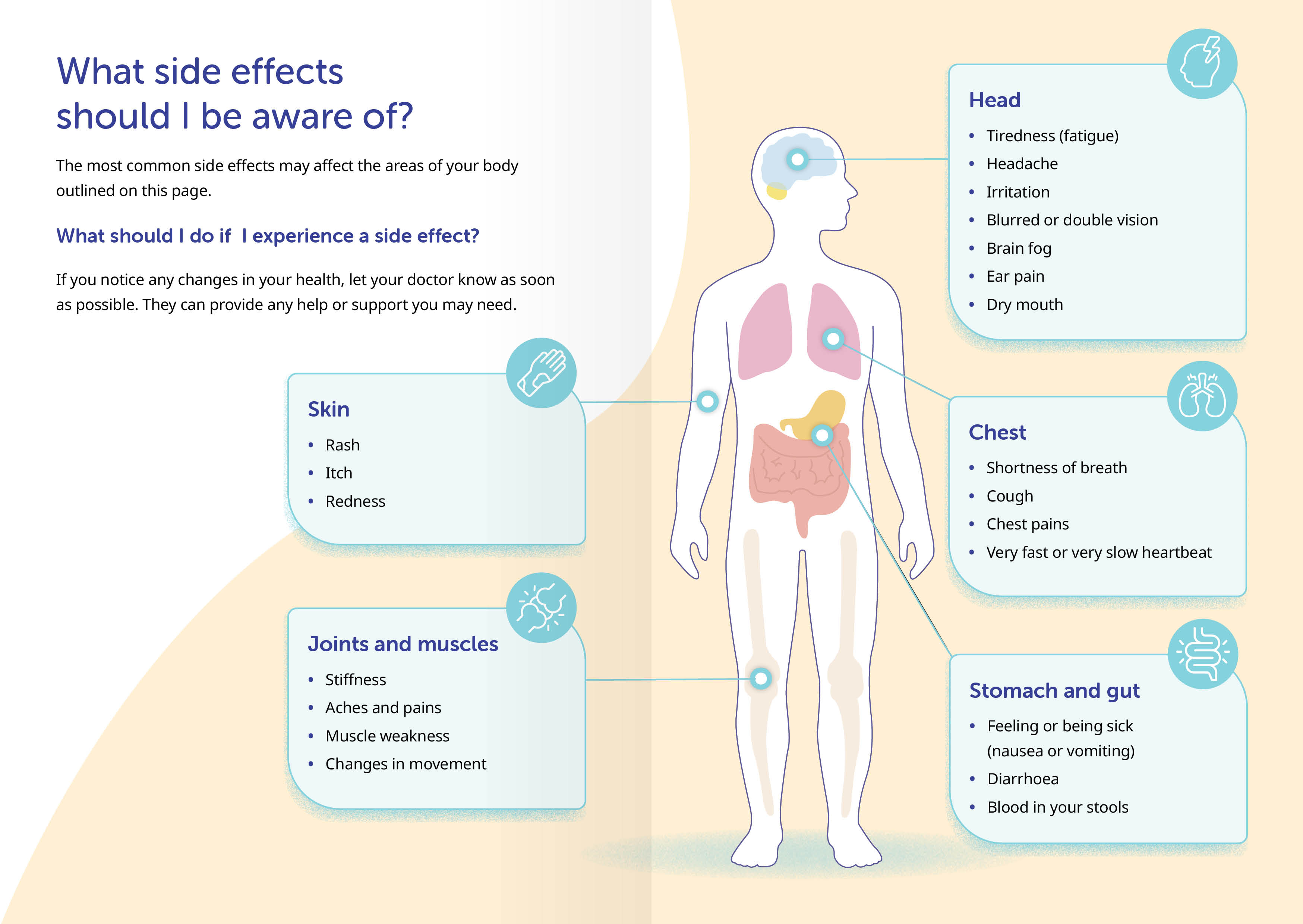
First, we translated all of the technical terms into lay language — “dry mouth” is just easier to process than “xerostomia”.
Next, we grouped everything by body system, so there’s no need to jump around the list for all related side effects.
Finally, we visualised each category on a silhouette, so readers know, at a glance, which body areas might be affected, and which specific changes to look out for.
By combining clear language and intuitive visuals, we’ve made potentially daunting side effect information easier to understand, recognise, and act on.
This content was provided by Cuttsy + Cuttsy
Company Details
Latest Content from Cuttsy + Cuttsy
Cuttsy+Cuttsy welcomes three new experts to the team
Cuttsy+Cuttsy is happy to be welcoming three new people to the team this month.
The gap between hype and reality, what patients really say about weight loss drugs
Explore real patient conversations about weight loss drugs, uncovering challenges, support strategies, and the critical need for clear, reliable healthcare communication.
Why patient-centricity depends on site-centricity in clinical trials
Explore why patient-centricity in clinical trials relies on site-centricity, emphasising the need to design trials that empower site teams to better support patients.
From cloudy to clear: visualising complex data privacy information
Discover how clear communication principles can transform complex clinical research topics, as we conclude our Health Literacy series with a practical example on participant data transparency.
UK clinical trial regs are changing — here’s what’s happening
Discover the transformative updates to UK clinical trial regulations, effective from April 2026, and learn how they aim to enhance transparency, participant safety, and trial efficiency.
The invisible influence on trial retention: why caregivers must be part of the plan
Caregivers are vital to clinical trial retention, yet often overlooked—this article highlights their challenges, impact, and how to better support them.
Where it all began: why we wrote ‘The Site Factor’
Discover the story behind ‘The Site Factor’ and how understanding site teams’ experiences can transform clinical trials for participants and researchers alike.
From text to context: Helping patients see side effects clearly
Discover how thoughtful design, clear language, and structured visuals can transform overwhelming side effect information into accessible, actionable insights for patients in our latest Health Literacy series.
Why patient engagement must come first in rare disease trials
Explore why patient engagement is the cornerstone of successful rare disease trials, emphasising the importance of designing studies around the realities of patients' lives to build trust and improve outcomes.
New Cuttsy+Cuttsy research shows why site support matters more than ever
Cuttsy+Cuttsy's upcoming report, 'The site factor: Where protocol meets practice in clinical trials', delves into the critical role of site staff in clinical research, offering insights into their daily challenges,...





